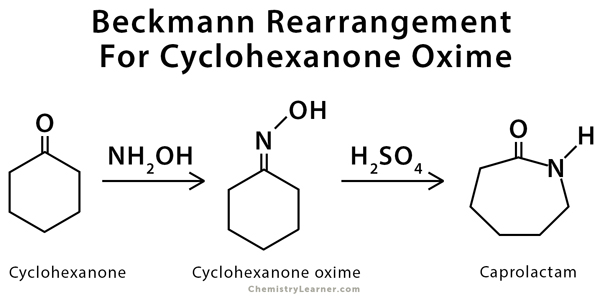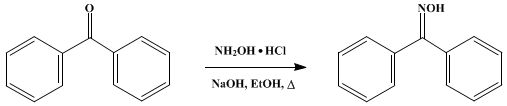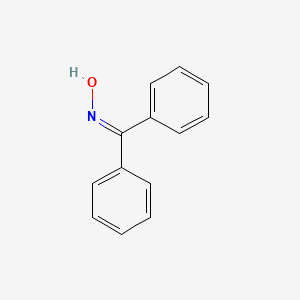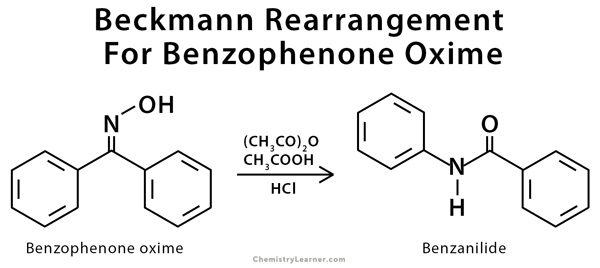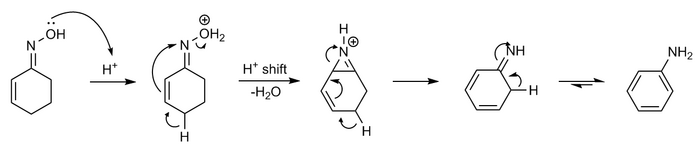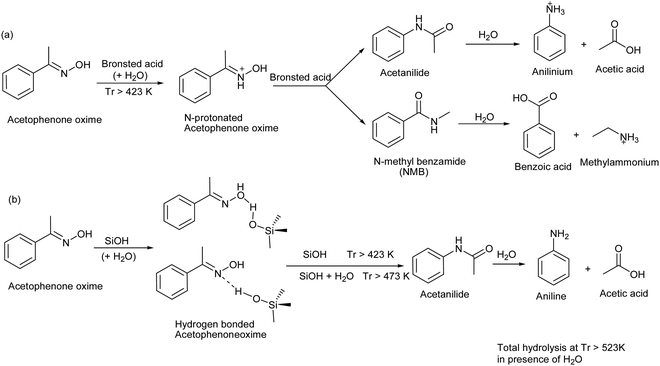
Beckmann rearrangement catalysis: a review of recent advances - New Journal of Chemistry (RSC Publishing) DOI:10.1039/D0NJ02034F
![Cyclohexanone Oxime Synthesis Notes - Xavier 1 of 5 Reminder: These notes are meant to supplement, not replace, the laboratory manual. Cyclohexanone Oxime Synthesis Notes Application: - [PDF Document] Cyclohexanone Oxime Synthesis Notes - Xavier 1 of 5 Reminder: These notes are meant to supplement, not replace, the laboratory manual. Cyclohexanone Oxime Synthesis Notes Application: - [PDF Document]](https://demo.fdocuments.in/img/378x509/reader024/reader/2021010102/5b015afb7f8b9af1148e02a7/r-1.jpg)
Cyclohexanone Oxime Synthesis Notes - Xavier 1 of 5 Reminder: These notes are meant to supplement, not replace, the laboratory manual. Cyclohexanone Oxime Synthesis Notes Application: - [PDF Document]
View of USE OF ALUMINA IN PREPARATION OF OXIME: A PRECURSOR FOR BECKMANN REARRANGEMENT | International Journal of Current Pharmaceutical Research

Synthesis, Herbicidal Evaluation, and Structure-Activity Relationship of Benzophenone Oxime Ether Derivatives

Cross‐Dehydrogenative C−O Coupling of Oximes with Acetonitrile, Ketones and Esters - Chen - 2019 - Advanced Synthesis & Catalysis - Wiley Online Library
Synthesis, herbicidal evaluation, and structure-activity relationship of benzophenone oxime ether derivatives - Document - Gale Academic OneFile

Oxime Formation - Oxime Formation This reaction is one example of the many derivatives which can be formed from carbonyl containing molecules Reaction | Course Hero

Cannizzaro Reaction - CANNIZZARO REACTION1 CHEMICAL REACTION O O H 2 OH N a OH B e n za ld e hy d e B e n zy l a lc oh
Oxime formation from hydroxylamine and ketone: a (computational) reality check on stage one of the mechanism. | Henry Rzepa's Blog
![Report: [3,3]-Rearrangements of O-Vinyl Oximes: Stereoselective Synthesis of 1,4-Dicarbonyl Compounds (56th Annual Report on Research Under Sponsorship of The American Chemical Society Petroleum Research Fund) Report: [3,3]-Rearrangements of O-Vinyl Oximes: Stereoselective Synthesis of 1,4-Dicarbonyl Compounds (56th Annual Report on Research Under Sponsorship of The American Chemical Society Petroleum Research Fund)](https://acswebcontent.acs.org/prfar/2011/images/Paper_11567_abstract_15187_0.gif)
Report: [3,3]-Rearrangements of O-Vinyl Oximes: Stereoselective Synthesis of 1,4-Dicarbonyl Compounds (56th Annual Report on Research Under Sponsorship of The American Chemical Society Petroleum Research Fund)