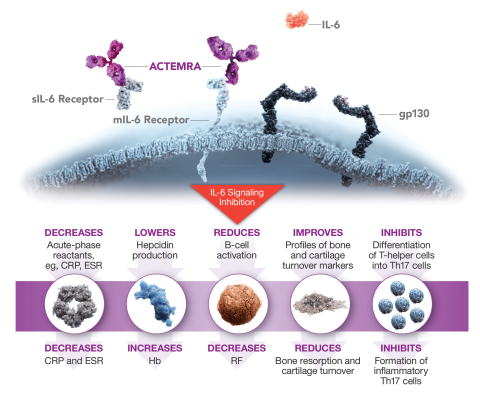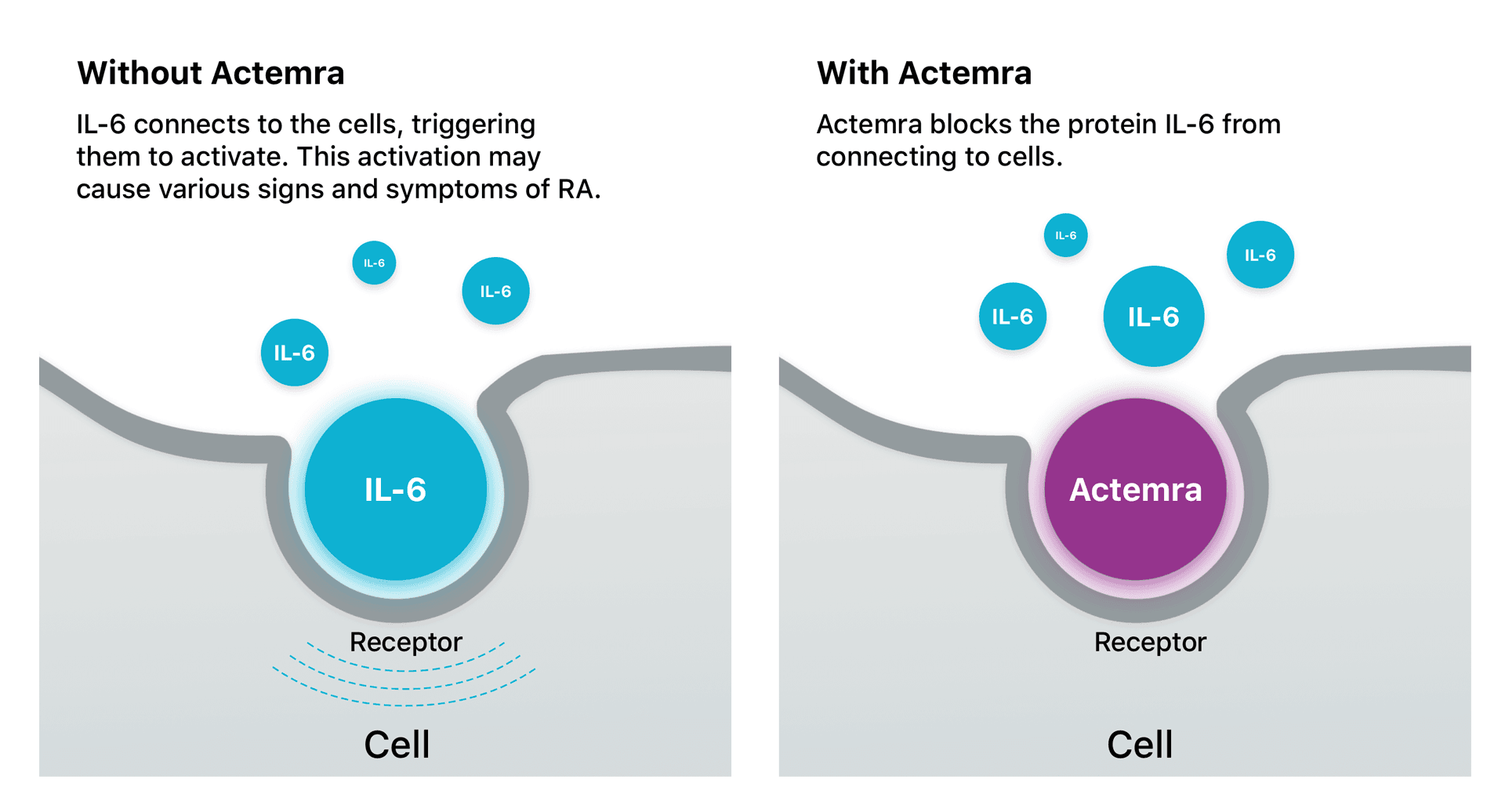
Subcutaneous formulation of tocilizumab for treatment of rheumatoid arthritis | Therapeutic Delivery
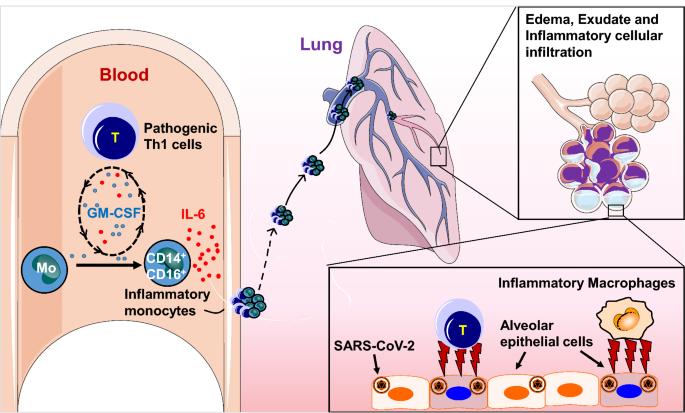
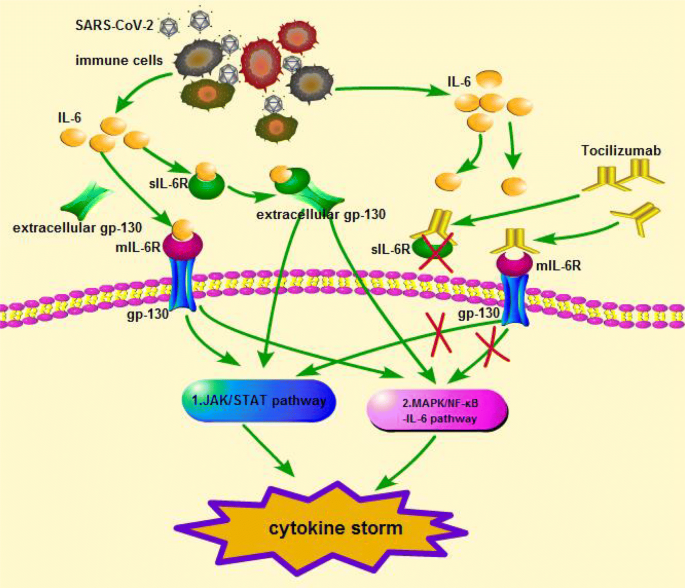
Why tocilizumab could be an effective treatment for severe COVID-19? | Journal of Translational Medicine | Full Text
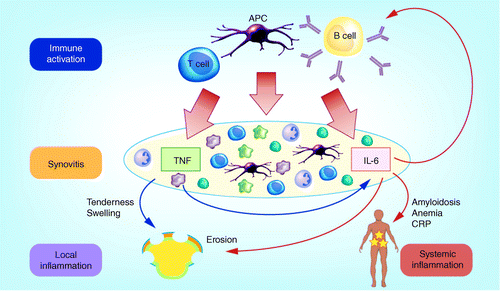
Subcutaneous formulation of tocilizumab for treatment of rheumatoid arthritis | Therapeutic Delivery

Risk of reactivation of hepatitis B virus (HBV) and tuberculosis (TB) and complications of hepatitis C virus (HCV) following Tocilizumab therapy: A systematic review to inform risk assessment in the COVID era
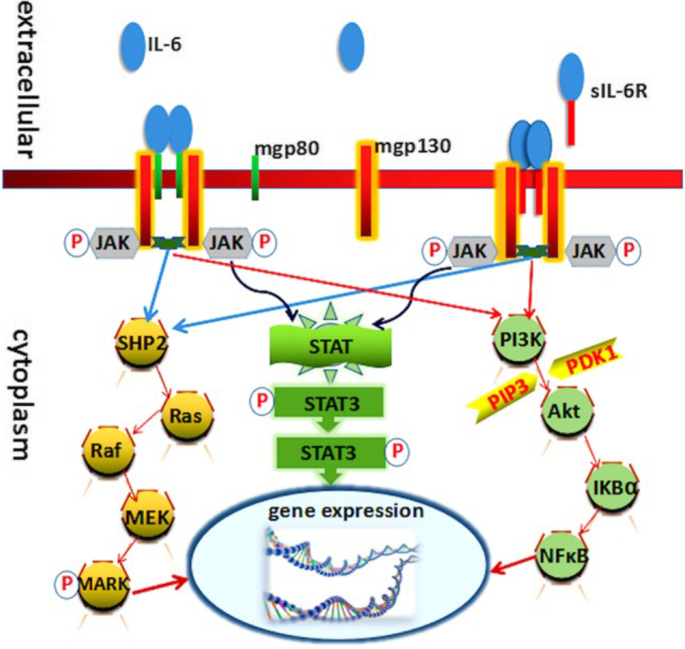
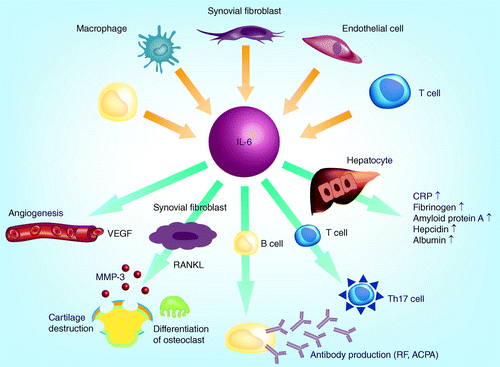
Interleukin-6 inhibition in the management of non-infectious uveitis and beyond | Journal of Ophthalmic Inflammation and Infection | Full Text

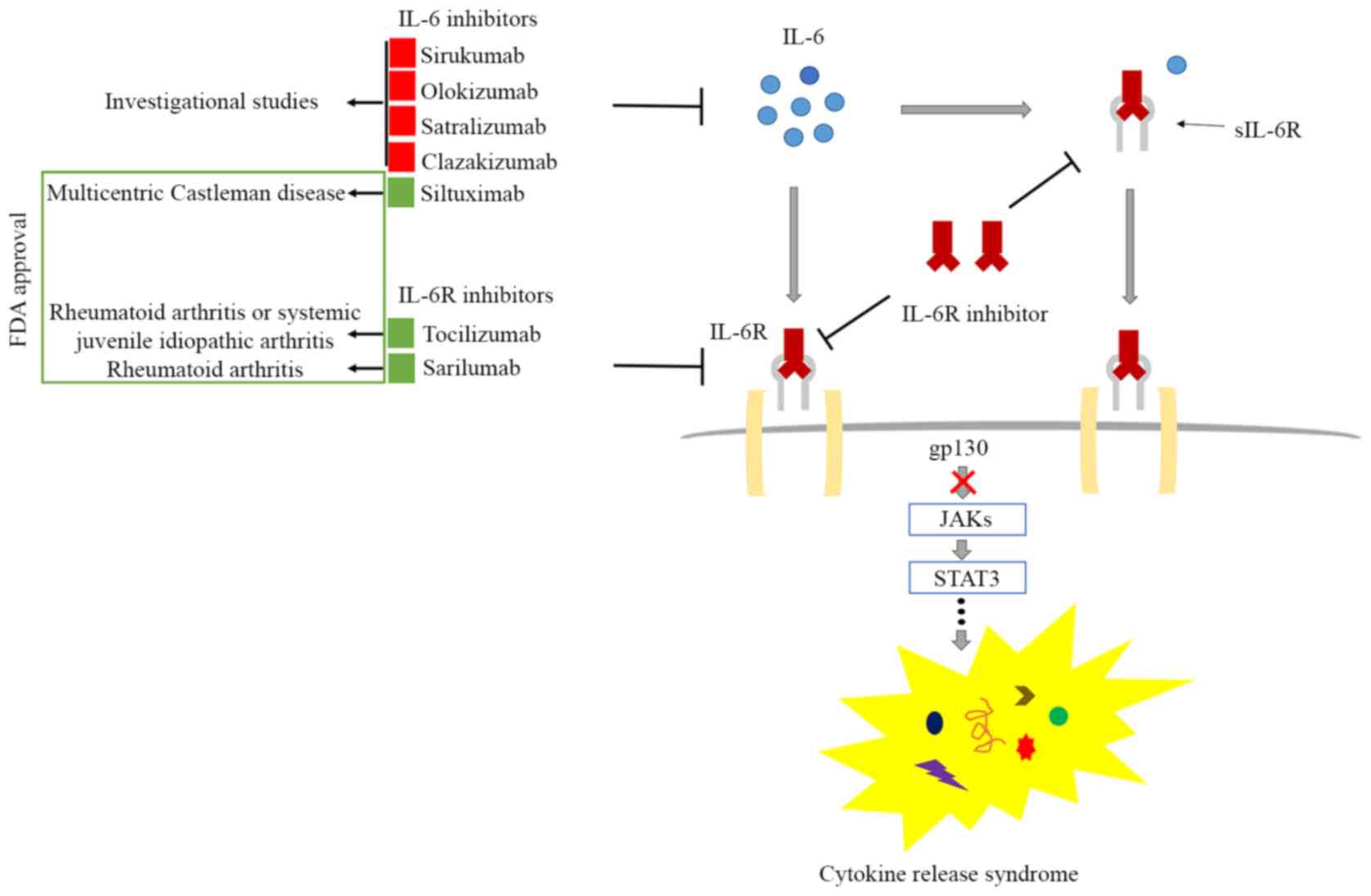
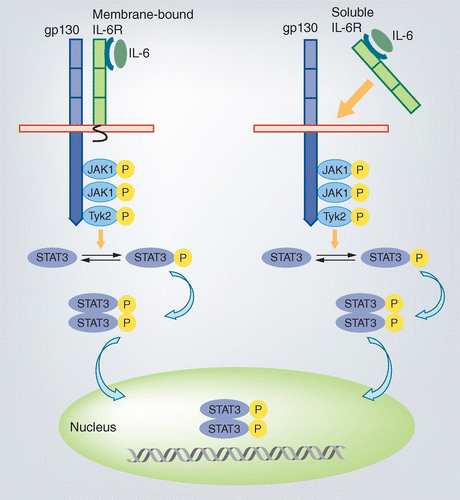
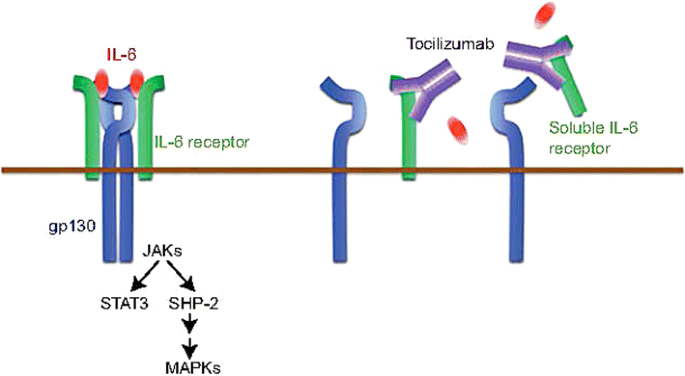
Anti‐interleukin‐6 receptor antibody, tocilizumab, for the treatment of autoimmune diseases - Tanaka - 2011 - FEBS Letters - Wiley Online Library

Clinical experience of IL-6 blockade in rheumatic diseases—Implications on IL-6 biology and disease pathogenesis - ScienceDirect

Effect of tocilizumab on clinical outcomes at 15 days in patients with severe or critical coronavirus disease 2019: randomised controlled trial | The BMJ
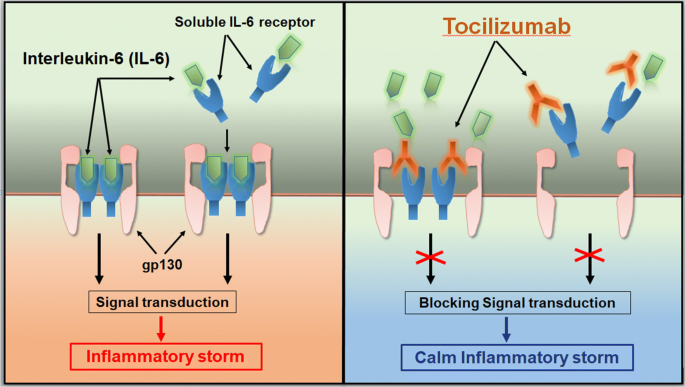
Why tocilizumab could be an effective treatment for severe COVID-19? | Journal of Translational Medicine | Full Text
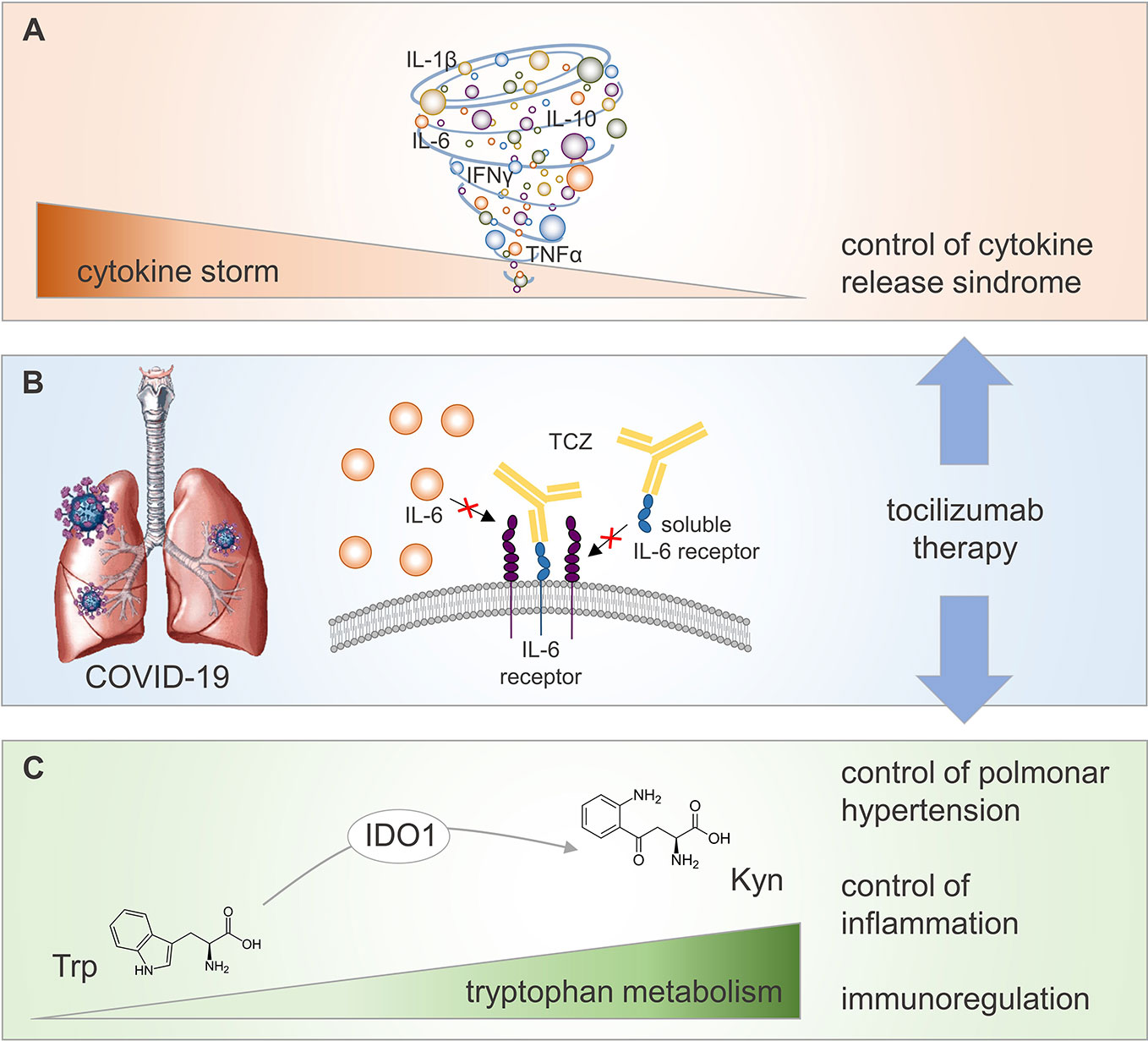
Frontiers | Potential Benefits of Tryptophan Metabolism to the Efficacy of Tocilizumab in COVID-19 | Pharmacology
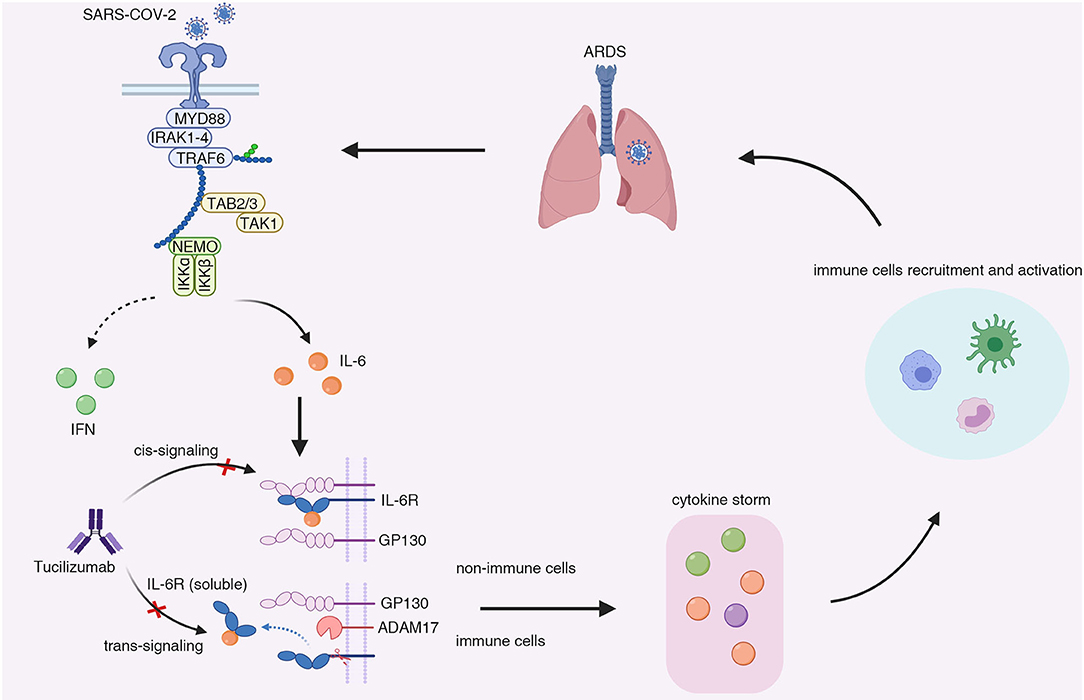
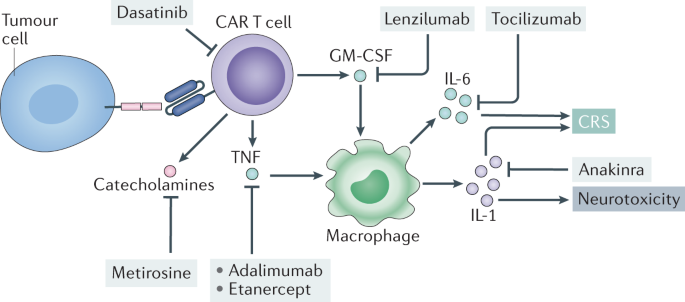
Frontiers | Tocilizumab: The Key to Stop Coronavirus Disease 2019 (COVID-19)-Induced Cytokine Release Syndrome (CRS)? | Medicine